- Buildings & Rooms
Rebecca Smith
Rotationsreinigung – Was ist das und welche Vorteile hat sie?
Warum muss ein Reinraum gereinigt werden? Was ist Rotationsreinigung, und wird sie wirklich gebraucht? Warum gibt es so viele verschiedene Chemikalien und wie funktionieren sie? Welche davon sollten verwendet werden? Als Beauftragte für Business Development eines Anbieters für Reinraum-Verbrauchsmaterial hat Rebecca Smith täglich mit solchen Fragen zu tun.
Um diese Fragen zu beantworten, hat sie mit Fachleuten gesprochen, eine Fülle von Informationen über Resistenzen von Mikroorganismen und die Wirkungsweisen von Desinfektionsmitteln gesichtet und die Anforderungen an die Sauberkeit in den verschiedenen Reinraumklassen studiert.
Den Reinraum reinigen
Die Antwort auf die erste Frage lautet: Natürlich müssen Reinräume gereinigt werden. Er mag zwar sauber aussehen, aber die meisten zu entfernenden Partikel sind mit bloßem Auge nicht erkennbar. Mit der Zeit lagern sich Schmutzpartikel, Zellreste und andere Verunreinigungen auf den Oberflächen des Reinraums ab und müssen entfernt werden. Die Norm DIN EN ISO 14644-1 definiert einen Reinraum wie folgt:
„Raum, in dem die Konzentration luftgetragener Partikel geregelt wird, der so konstruiert und verwendet wird, dass die Anzahl der in den Raum eingeschleppten bzw. im Raum entstehenden und abgelagerten Partikel kleinstmöglich ist, und in dem andere reinheitsrelevante Parameter wie Temperatur, Feuchte und Druck nach Bedarf geregelt werden“ [10].
Durch Reinigen werden die Ablagerung von Partikeln im Reinraum reduziert. Als beste Technik zum Reinigen einer Oberfläche in einem Reinraum gilt das feuchte Reinigen. Dazu werden im Allgemeinen Tücher verwendet, die mit einer Desinfektions- oder Reinigungsmittellösung getränkt sind. Beim mechanischen Abwischen wird eine bestimmte Menge von Partikeln von der Oberfläche entfernt. Sind das Tuch und die Oberfläche feucht, wird die Haftung zwischen den Partikeln und der Oberfläche stärker verringert und es können mehr Partikel entfernt werden.
Abtöten von Mikroorganismen und Gute Herstellungspraxis gemäß EU-Leitlinien
Was ist eigentlich Rotationsreinigung? Sie bezieht sich auf die Keimbelastung, also die Anzahl der Mikroorganismen, die auf einer unsterilisierten Oberfläche leben. Mit der Rotationsreinigung sollen nicht nur Schmutz, Fusseln, Zellreste usw. entfernt werden, sondern es soll auch die Kontamination in Form eventueller lebender Organismen abgetötet werden. Beim Reinigen eines Reinraums wird zweifellos auch ein Teil der mikrobiellen Besiedelung entfernt. Es ist jedoch unwahrscheinlich, dass alle „erwischt“ werden, daher müssen die nicht entfernten Mikroorganismen abgetötet werden.
Um die Keimbelastung des Reinraums zu senken, wird Desinfektionsmittel benötigt, also Chemikalien, die Mikroorganismen abtöten können. Dabei müssen vermutlich zwei oder mehr der Mittel abwechselnd benutzt werden, um alle Keime abzutöten. Diesen Turnus nennt man Rotation, daher der Begriff Rotationsreinigung.
In der Leitlinie für Gute Herstellungspraxis für Human- und Tierarzneimittel, (EudraLex Band 4), Anhang 1 über die Herstellung steriler Arzneimittel, stellt die EU in Punkt 61 fest:
„Die Dekontaminierung sauberer Bereiche ist besonders wichtig. Sie sollten nach einem schriftlich festgelegten Programm gründlich gereinigt werden. Bei Verwendung von Desinfektionsmitteln sollte mehr als eine Sorte verwendet werden. Der Bereich sollte regelmäßig auf die Entwicklung resistenter Stämme überwacht werden.” [6]
Warum muss mehr als nur ein Desinfektionsmittel verwendet werden? Damit es nicht zu Resistenzen kommt. Es gibt zwei Arten von Resistenz – natürlich vorkommende Resistenz und Bildung resistenter Stämme durch Selektion.
Bildung resistenter Stämme durch Selektion
Bei Organismen wie MRSA konnte verfolgt werden, wie die Entwicklung genetischer Resistenz funktioniert. Ein Bakterium, das zunächst noch durch Methicillin abgetötet werden konnte, entwickelte mit der Zeit eine genetische Resistenz, und dieses Antibiotikum wirkt nun nicht mehr dagegen. Es wird angenommen, dass dasselbe mit der Wirksamkeit von Desinfektionsmitteln gegen Mikroorganismen im Reinraum passieren könnte.
Mit der Zeit könnten beispielsweise Bakterien, die bisher empfindlich gegen Alkohole waren, eine genetische Resistenz gegen solche Mittel entwickeln, sodass diese Desinfektion nicht mehr wirkt. Aus diesem Grund erscheint es ratsam, diesem Effekt durch die Verwendung mehrerer Desinfektionsmittel vorzubeugen. Bisher liegen zwar noch keine Anhaltspunkte vor, dass so etwas passiert oder auch nur möglich ist, aber Vorsichtsmaßnahmen sollten trotzdem getroffen werden.
Die Bedingungen, die für die Entwicklung von MRSA ausschlaggebend waren, und die Bedingungen in einem Reinraum unterscheiden sich allerdings. Damit sich Resistenz entwickelt, müssten, vereinfacht gesagt, einige wenige Bakterien die Anwendung eines bestimmten gegen sie wirksamen Mittels überleben. Diese können sich nun vermehren und den Vorteil, den sie gegenüber anderen Stämmen haben und der ihr Überleben ermöglicht hat, weitergeben. Der überlebende Stamm wächst und gedeiht.
Beim nächsten Reinigen überleben wieder nur wenige von diesen Bakterien, vermehren sich wieder, und der Überlebensvorteil wird weitergegeben und wird immer stärker. Wird immer wieder dasselbe Mittel verwendet, wiederholt sich dieser Vorgang vielfach. Der Vorteil wird immer größer, bis der Stamm schließlich komplett resistent gegen das Mittel ist. Da in Reinraumumgebungen meist übermäßig häufig und mit sehr starken Mitteln gereinigt wird, überleben nur sehr wenige Mikroorganismen. Deshalb ist es unwahrscheinlich, dass per Selektion resistente Stämme entstehen. Auch besitzen Antibiotika eine sehr spezifische und gezielte Wirkungsweise, durch die eine Selektion wahrscheinlicher wird. Desinfektionsmittel dagegen haben ein breites Wirkungsspektrum, sodass eine Selektion weniger wahrscheinlich ist.
Die Bildung resistenter Stämme durch Selektion ist ein allmählicher Entwicklungsprozess. Die Resistenz ist keine ursprüngliche Eigenschaft des Organismus. Dies unterscheidet diese Art der Resistenz von der natürlich vorkommenden Resistenz.
Natürlich vorkommende Resistenz
Natürlich vorkommende Resistenz beruht auf der Tatsache, dass verschiedene Desinfektionsmittel auf verschiedene Art und Weise wirken. Nicht alle sind gleich wirksam gegen alle Organismen. Manche sind sehr wirksam gegen Bakterien, aber nicht gegen Pilze, andere sind wirksam gegen Viren, nicht aber gegen Endosporen.
Aufgrund der verschiedenen Wirkungsweisen von Desinfektionsmitteln sind manche Mikroorganismen von Natur aus besser gegen sie gewappnet. Diese Eigenschaft ist nicht erlernt, selektiert oder genetisch vererbt, sondern liegt im Aufbau des Mikroorganismus selbst begründet. Ein Vergleich macht dies klar: Dass es große Menschen von Natur aus leichter beim Basketball haben als kleine, ist weder erlernt noch antrainiert. Der Vorteil liegt schlicht in ihrer Körpergröße begründet.
Wirkungsweisen von Desinfektionsmitteln
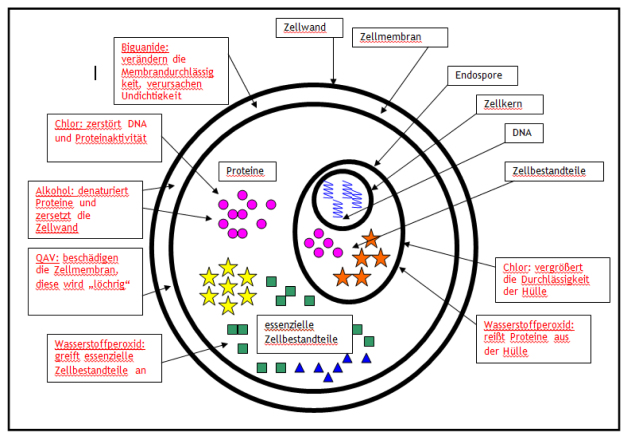
Desinfektionsmittel auf Alkoholbasis sind gegen die meisten Mikroorganismen wirksam, nicht jedoch gegen Endosporen. Sie wirken durch Denaturierung von Proteinen in der Zelle, wodurch diese verklumpen und funktionslos werden. In der Zellwand führt dies zum Strukturverlust und Zusammenbruch.
Quartäre Ammoniumverbindungen (QAV oder Quats) wirken durch Beschädigung der Zellmembran, wodurch Bestandteile des Zellinnern austreten und sich zersetzen. Sie wirken gegen Bakterien, behüllte Viren und Pilze, aber kaum gegen unbehüllte Viren und Endosporen.
Biguanide verändern die Durchlässigkeit der Zellmembran. Sie beschädigen die äußeren Schichten und greifen die inneren Schichten an. Auch dies führt zum Austreten von Zellbestandteilen. Die Wirkung ist ähnlich wie die der QAV.
Chlor ist ein starkes Oxidationsmittel. Es oxidiert die DNA und Zellproteine und zerstört dadurch ihre Aktivität. Chlorhaltige Desinfektionsmittel töten bei höheren Konzentrationen fast alles ab, einschließlich Endosporen.
Wasserstoffperoxid ist hoch reaktiv und wirkt oxidierend, wobei freie Hydroxylradikale entstehen. Diese greifen dann die essenziellen Zellbestandteile an. Desinfektionsmittel auf Basis von Wasserstoffperoxid töten alles einschließlich Endosporen ab, können aber auch die zu reinigenden Oberflächen angreifen.
Aus der Wirkungsweise von Biguaniden auf die Zellwand und die Zellmembran folgt also auch, dass solche Mittel nicht sehr wirksam gegen Mikroorganismen mit einer sehr starken Zellwand sind. Solche Mikroorganismen haben eine natürliche Resistenz gegen die Wirkung von Biguaniden.
Endosporen
Endosporen sind extrem schwierig abzutöten. Die Endospore ist ein Stadium eines Bakteriums oder Virus, das dieses bei ungünstigen äußeren Bedingungen annimmt, zum Beispiel bei Nährstoffmangel, Wassermangel, Temperatur- oder pH-Änderungen. Um den Zellkern und die essenziellen Elemente herum bildet sich eine extrem robuste Hülle. Dieses Stadium überdauert lange Phasen, bis wieder günstigere Bedingungen eintreten. Dann wird die Hülle durchlässig und die Zelle wieder aktiv.
Aufgrund dieser starken Hülle ist die Endospore sehr schwer abzutöten; sie widersteht Gammastrahlung und der Wirkung vieler Desinfektionsmittel. Desinfektionsmittel auf Chlor- oder Wasserstoffperoxidbasis wirken gegen Endosporen und werden daher oft als sporizid bezeichnet. Chlor erhöht die Durchlässigkeit der Endosporenhülle, und Wasserstoffperoxid kann dort Proteine herausbrechen.
Wirkungsspektrum
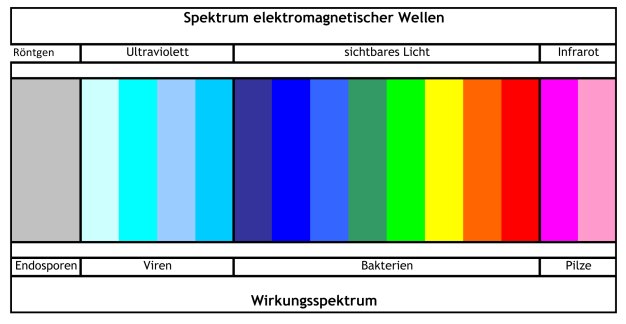
Mit der Verwendung verschiedener Desinfektionsmittel kann das Wirkungsspektrum erhöht werden, also der Anteil der Mikroorganismen, der abgetötet werden kann. Das Wirkungsspektrum kann ähnlich dargestellt werden wie das Spektrum elektromagnetischer Wellen. Wenn nur das sichtbare Licht betrachtet wird, wird ein sehr großer Anteil des Spektrums außer Acht gelassen, da es zusätzlich noch Röntgen- und Ultraviolettstrahlung gibt. Ähnlich wäre es, wenn immer nur Desinfektionsmittel verwendet werden würden, die Bakterien abtöten können. So würde nichts gegen Endosporen und die anderen Mikroorganismen unternommen werden. Bei der Auswahl von Desinfektionsmitteln sollten daher jene berücksichtigt werden, welche einen größtmöglichen Anteil des Spektrums abdecken.
Wenn ausschließlich Mittel verwendet werden, welche Bakterien, nicht aber Viren abtöten, werden zusätzlich noch günstigere Bedingungen für die Viren geschaffen. In einer für Viren derart günstigen Umgebung wirkt auch das speziell ausgewählte Desinfektionsmittel nicht mehr gegen sie.
Auswahl eines Desinfektionsmittels
Welche Desinfektionsmittel sollten verwendet werden? Es wurde deutlich gemacht, dass die Verwendung eines Sporizids sehr wichtig ist, aber sporizid wirksame Desinfektionsmittel sind im Allgemeinen so scharf, dass sie nicht täglich eingesetzt werden können. Aus diesem Grund wird der Einsatz des Sporizids abwechselnd mit einem anderen wirksamen Desinfektionsmittel empfohlen, welches sich für die regelmäßige Verwendung besser eignet. Zusätzlich wäre die Verwendung eines alkoholhaltigen Mittels zu empfehlen, da diese gut gegen die meisten Keime wirken und auch eventuelle Reste anderer Desinfektionsmittel gut entfernen.
Zusammenfassung
Reinräume müssen gereinigt werden, um gemäß der Norm DIN EN ISO 14644-1 die Ablagerung von Partikeln zu minimieren. Bei der Rotationsreinigung werden abwechselnd verschiedene Desinfektionsmittel verwendet, um die Keimbelastung im Reinraum zu senken.
Die EU-GHP-Leitlinien empfehlen eine gründliche Reinigung, ein schriftlich festgelegtes Reinigungsprogramm und, bei Verwendung von Desinfektionsmitteln, den Einsatz von mehr als einem. Der Grund für diese Mühen ist die Vermeidung von Resistenzen. Darunter fallen natürlich vorkommende Resistenzen, durch die Mikroorganismen gegen ein bestimmtes Desinfektionsmittel schlicht unempfindlich sind, und resistente Stämme, die sich durch Selektion aus ehemals empfindlichen Keimen entwickeln könnten.
Es sollten verschiedene Arten von Desinfektionsmitteln verwendet werden, da deren Wirkstoffe sich in ihrer Wirkungsweise unterscheiden. Sie wirken gegen verschiedene Arten von Keimen, haben daher also ein breiteres Wirkungsspektrum. Welche Desinfektionsmittel wie häufig verwendet werden sollten, ist abhängig von vielen Faktoren, zum Beispiel vom Reinigungsprozess selbst, der Reinraumklasse, der Art der Verschmutzung, den Darreichungsformen der Mittel, der Benutzerfreundlichkeit und Umweltverträglichkeit. Als Leitfaden kann gelten: abwechseln zwischen drei Mitteln, und zwar einem alkoholhaltigen, einem anderen allgemeinen Desinfektionsmittel und einem Sporizid.
Quellen:
1. Sandle, T. (2012) A guide to cleaning & disinfecting cleanrooms, Surrey: Grosvenor House Publishing.
2. Whyte, W. (2010) Cleanroom Technology, Fundamentals of design, testing and operation, West Sussex: John Wiley & Sons Ltd.
3. Araújo, P. Lemos, M. Mergulhão. Melo, L. Simões, M. (2011) Antimicrobial resistance to disinfectants in bio-films, Science against microbial pathogens: communicating current research and technological advances, p826.
4. Sartain, E. (2005) Disinfectant Rotation, Available: www.cemag.us/print/articles/2005/03/disinfectant-rotation.
5. Martinez, J.E. (2009) The rotation of disinfectants principle: true or false? Available: http://www.pharmtech.com/pharmtech/Article/The-Rotation-of-Disinfectants-Principle-True-or-Fa/ArticleStandard/Article/detail/580032.
6. "Annex 1: Manufacture of Sterile Medicinal Products," Good Manufacturing Practice (GMP) Guidelines (Brussels, Nov, 2008), Available: http://ec.europa.eu/health/files/eudralex/vol-4/2008_11_25_gmp-an1_en.pdf.
7. McDonnell, G. Denver Russell, A. (1999) Antiseptics and disinfectants: activity, action and resistance, Clinical Microbiology Reviews, Jan 1999, vol 12. No 1 147-179.
8. Critical Cleaning Bulletin (2007) contact Weitzel, S. Critical Process Cleaning, CANI, Inc, Available: http://cdn.shopify.com/s/files/1/0186/2832/files/BULLETIN_selection_and_rotation_of_disinfectants.pdf?380.
9. Guideline for disinfectant and sterilization in healthcare facilities, 2008, Centers for Disease Control and Prevention, Available: http://www.cdc.gov/hicpac/disinfection_sterilization/6_0disinfection.html.
10. “Part 1: Classification of air cleanliness” Cleanrooms and associated controlled environments, The European Standard EN ISO 14644-1:1999.
________________________________________
Über die Autorin Rebecca Smith
Ihr wissenschaftliches Interesse führte sie zu einem Biologiestudium, das sie mit einem Bachelor-Grad abgeschlossen hat. Danach zog es sie in den Pharma-Sektor, zur Firma Merck Sharp and Dohme (MSD). Sie arbeitete 5 Jahre lang als Pharmavertreterin für MSD und konnte dabei wertvolle Erfahrungen sammeln, die auch für eine Tätigkeit in der Reinraumbranche unverzichtbar sind. Seit 2 Jahren ist sie bei Connect 2 Cleanrooms für den Bereich Business Development zuständig. In dieser Position unterstützt sie Reinraumanwender in der täglichen Arbeit mit ihren Reinräumen. In ihrer Freizeit entspannt sie sich bei der Erziehung ihres Labradors Charlie.
Connect 2 Cleanrooms
LA2 6RH Halton, Lancashire
United Kingdom